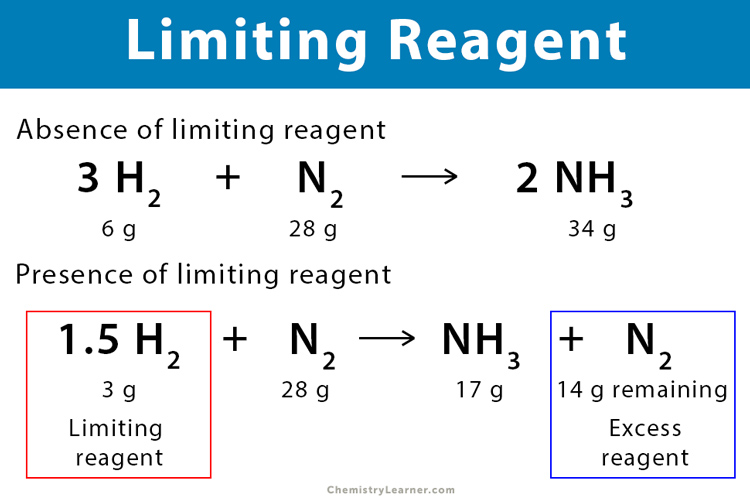
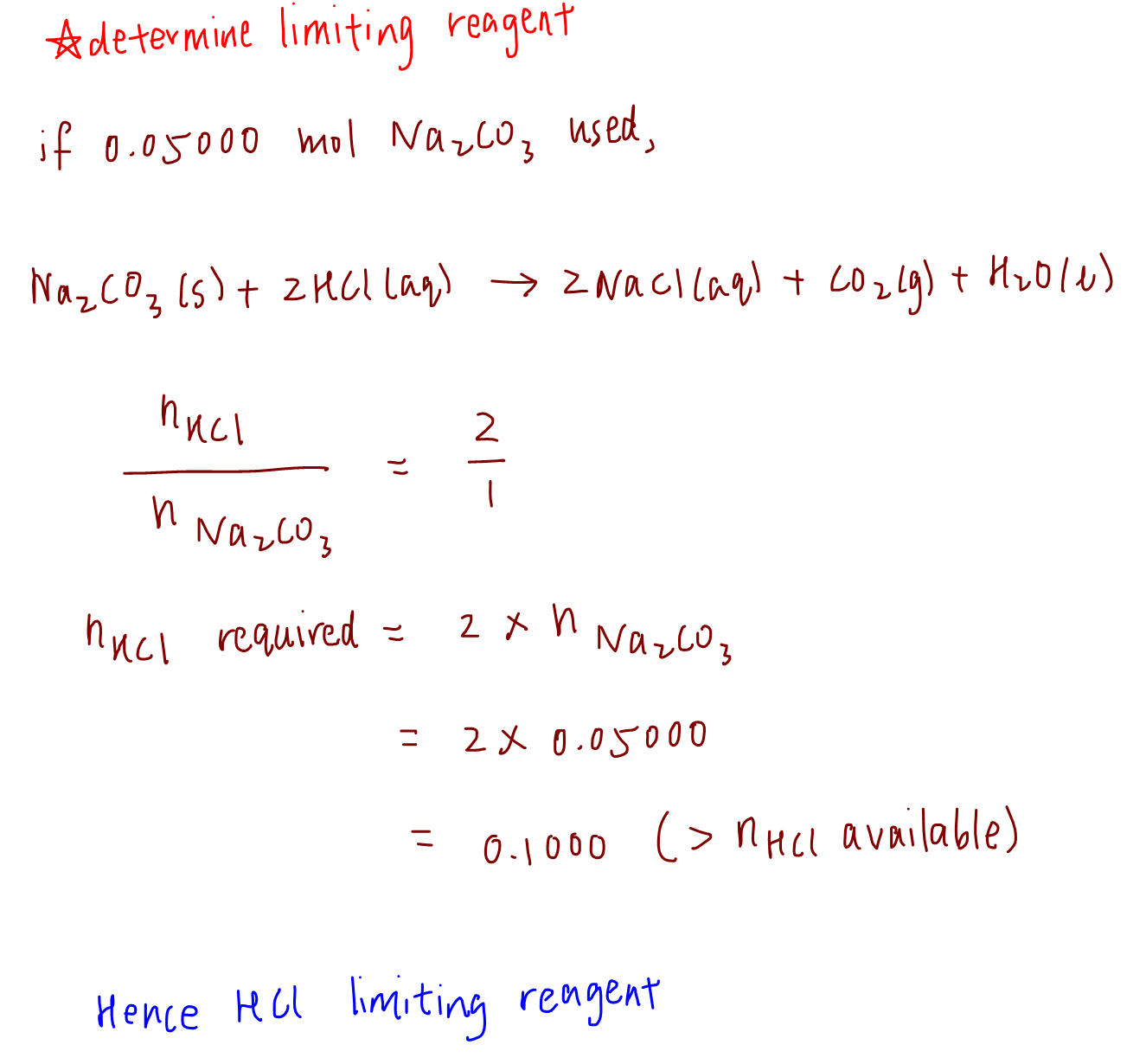SOLVED: Calculate the Limiting Reactant and Remaining Substances Given the amount of each reactant in the table below, determine the limiting reactant, the grams of copper metal created, and the amount of

stoichiometry - Why do we only take into account the first product while calculating a limiting reactant? - Chemistry Stack Exchange

CHEM 101: Dimensional Analysis Limiting Reagent, Theoretical Yield, Percent Yield, Excess Reactant 2 - YouTube